

Complete platform for precision medicine and next-generation sequencing (NGS).
Specialized genomics system that ensures total control of the analytical cycle, from sample reception to the release of molecular reports. Developed for laboratories operating with next-generation sequencing (NGS), molecular testing, and high-complexity genomic analysis.
Individualized control of biological material.
Comprehensive management from reception to final processing, with specific traceability for each type of genetic material:


Complete audit trail for genomics.
Unalterable historical record of all interventions in the analytical cycle:
Benchtop control.
Unified management of manual and automated stages:


Strict control over the most sensitive stages of the genomic workflow:


90% reduction in report preparation time.

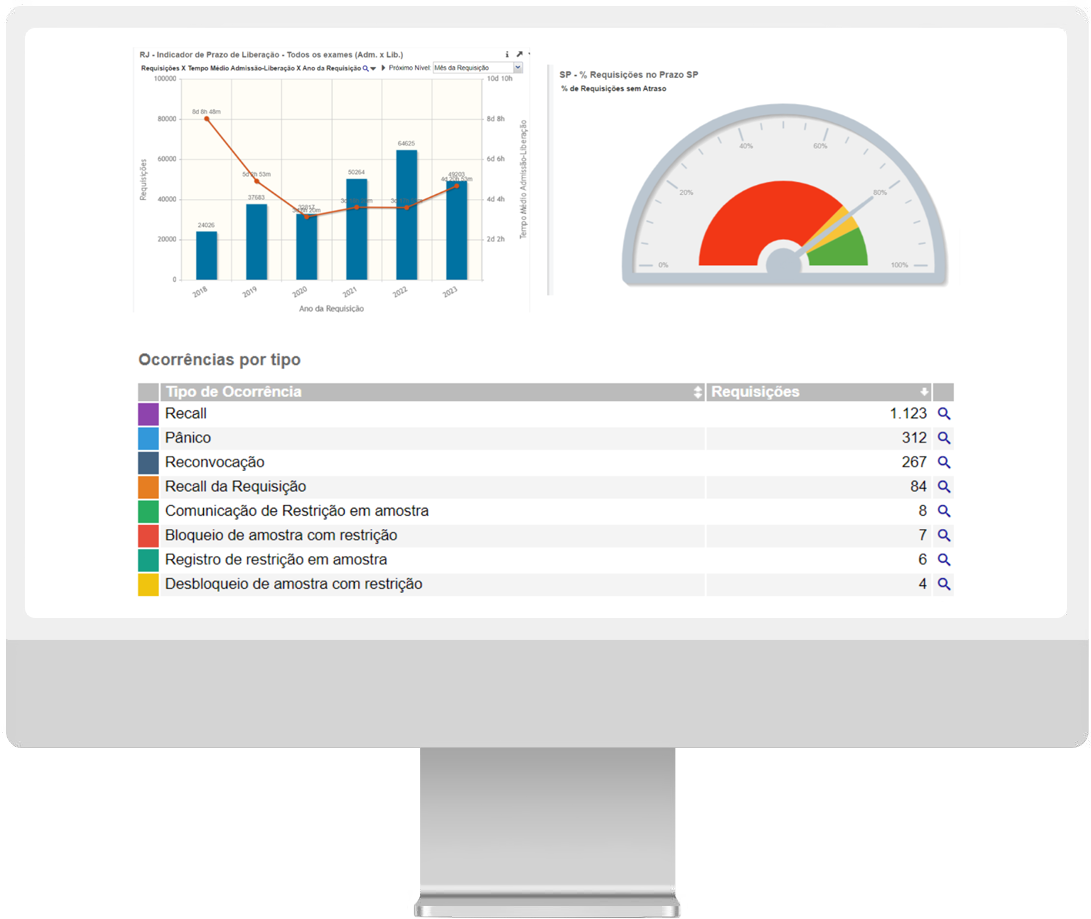
Business intelligence for genomic laboratories.
The foundation for genomics accreditations.
Architecture developed to meet the rigorous quality standards required in clinical genomics.



Proven Benefits.
Direct integration with bioinformatics.
Rigorous control of barcodes and sample sheets.
From the blood tube to the molecular report.From the blood tube to the molecular report.
Approved by national and international accreditations.
From PCR to whole-genome sequencing.
Optimization of NGS runs and reduction in reprocessing.
Discover how MotionLIS Genomics can transform your genomic laboratory’s operations.
Contact our team to schedule a personalized demo.